-
-

FLUORIDE
What Is Stannous Fluoride Toothpaste?Discover what is Stannous Fluoride Toothpaste and its importance to prevent cavities and other oral health problems.

TEETH WHITENING
Whitening toothpaste - hydrogen peroxide vs. carbamide peroxideIf you lose one or more of your front teeth due to injury or decay, you may feel ...
-
Science & InnovationOral Health Commitment
- Oral Health Commitment
- Bright Smiles, Bright Futures
- Educational Resources
- Mobile Dental Van
- Volunteer
- ORAL HEALTH CHECK
- PRODUCT MATCH
- Oral Health and Dental Care | Colgate®
- Oral Health
- Cleft Lip Causes, Drugs And Medications


Cleft lip is a birth defect characterized by a split or separation of the upper lip and can occur on one or both sides. While the exact cause is unknown, risks of having a baby with the physical deficiency may increase with the use of certain drugs during pregnancy. According to WebMD, oral cleft affects 700 babies each year and is the fourth most common birth defect in the U.S.
Topiramate and Oral Cleft
According to the American Dental Association (ADA), pregnant women who take the drug Topiramate increase the risks of oral cleft in babies. This conclusion stems from data the North American Antiepileptic Drug Pregnancy Registry collected, which the Food and Drug Administration (FDA) released in a safety alert. Risk extends to generic versions of Topiramate, as well.
Topiramate is a drug that treats epileptic seizures. The drug increases risks of cleft lip in newborns when its consumption occurs during the first trimester of pregnancy. In the article cited in the previous paragraph, the ADA explains that, "infants exposed to Topiramate as a single therapy experienced a 1.4 percent prevalence of oral clefts, compared with a prevalence of 0.38 percent - 0.55 percent in infants exposed to other antiepileptic drugs..."
HIV Drugs
Antiviral drugs taken by a pregnant woman may link to oral cleft in her baby, according to a recent American Dental Association news post. The study, published in the Cleft Palate-Craniofacial Journal, analyzes data in the FDA Adverse Events Reporting System (AERS) Database.
According to the study analysis, 26 cases of cleft lip and palate associate with antiretroviral therapies. The drugs included efavirenz, nelfinavir, nevirapine and lamivudine. The cases applied to both monotherapy and combination therapy situations for HIV-infected mothers.
The Conclusions
Cleft lip develops from a complex combination of genetic and environmental variables. The incidence of oral cleft in newborns may increase when pregnant women consume Topiramate and certain HIV drugs.
More research is necessary to solidify any direct links between the drugs and the facial defect that affects an infant's ability to eat, breathe and speak. To correct the defect, a baby usually goes through surgery at three or four months of age.
This article is intended to promote understanding of and knowledge about general oral health topics. It is not intended to be a substitute for professional advice, diagnosis or treatment. Always seek the advice of your dentist or other qualified healthcare provider with any questions you may have regarding a medical condition or treatment.
Related Articles

Cleft lip palate
What Is a Submucous Cleft Palate?Although most orofacial clefts are identified when the baby is born, a submucous cleft palate may not be diagnosed until later in his or her life. Learn more here.

Cleft lip palate
Cleft Lip Causes, Drugs And MedicationsCleft lip is a birth defect characterized by a separation of the upper lip and can occur on one or both sides. Find out more about drugs and risks for newborns.

Cleft lip palate
What Is Cleft Lip and Cleft Palate?A cleft lip is an upper lip that is split, something that affects one out of every 700 babies in the U.S. Find out more about cleft lip and cleft palate, here.

Cleft lip palate
How Does Your Child’s Cleft Palate Affect Their Oral Health?How does a cleft palate affect your child's oral health? What can you do to take care of their smile? Learn more about oral clefts and teeth, here.
Related Products
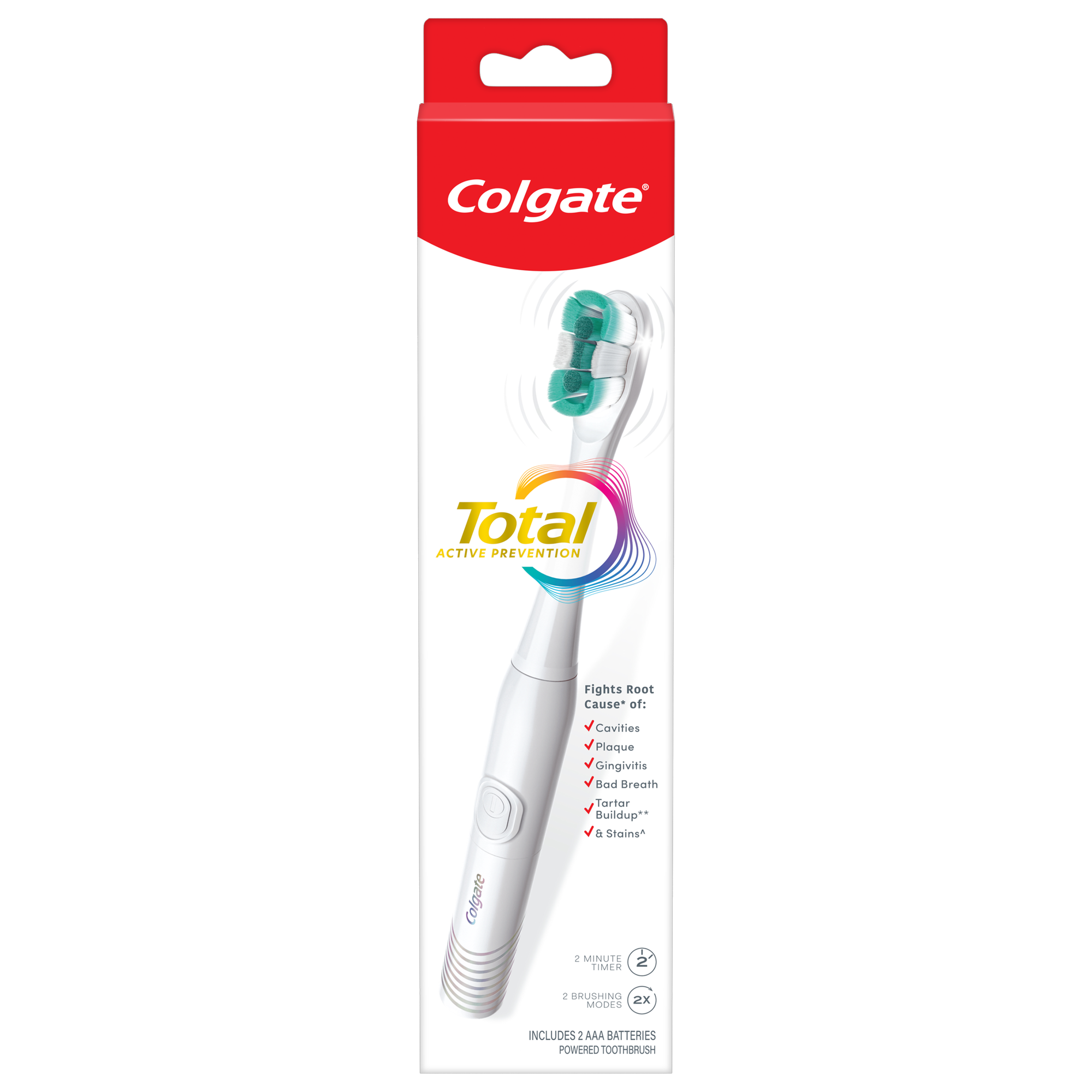
Power away plaque with Colgate Total Battery Powered Toothbrush. This battery operated toothbrush for adults fights the root cause* of cavities, plaque, gingivitis, bad breath, tartar buildup**, and stains***. Plus, this battery toothbrush has a built in 2 minute timer and features two cleaning modes, Sensitive and Regular, to cater to your unique oral care needs.

Colgate Total Alcohol Free* Gum Health Mouthwash delivers 24-hour protection** against bacteria and also helps prevent gum problems

Colgate Total Plaque Pro Release Fresh Mint Toothpaste dissolves and lifts away gum harming plaque with daily brushing.

Help prevent sensitivity from day 1* with Colgate Total Active Prevention Sensitivity Mineral Shield Mint Toothpaste. This Colgate Total Sensitivity Toothpaste with Fluoride is a patented antisensitivity toothpaste that fights the root cause** of gingivitis, plaque, tartar, cavities, sensitivity, bad breath, weak enamel, and stains. Colgate Total Sensitivity Mineral Shield Toothpaste helps prevent tooth sensitivity by creating a mineral shield around sensitive spots on your teeth.
*with twice daily use
**via protection against bacteria and dietary exposures, with daily brushing

Helping dental professionals
More professionals across the world trust Colgate. Find resources, products, and information to give your patients a healthier future




